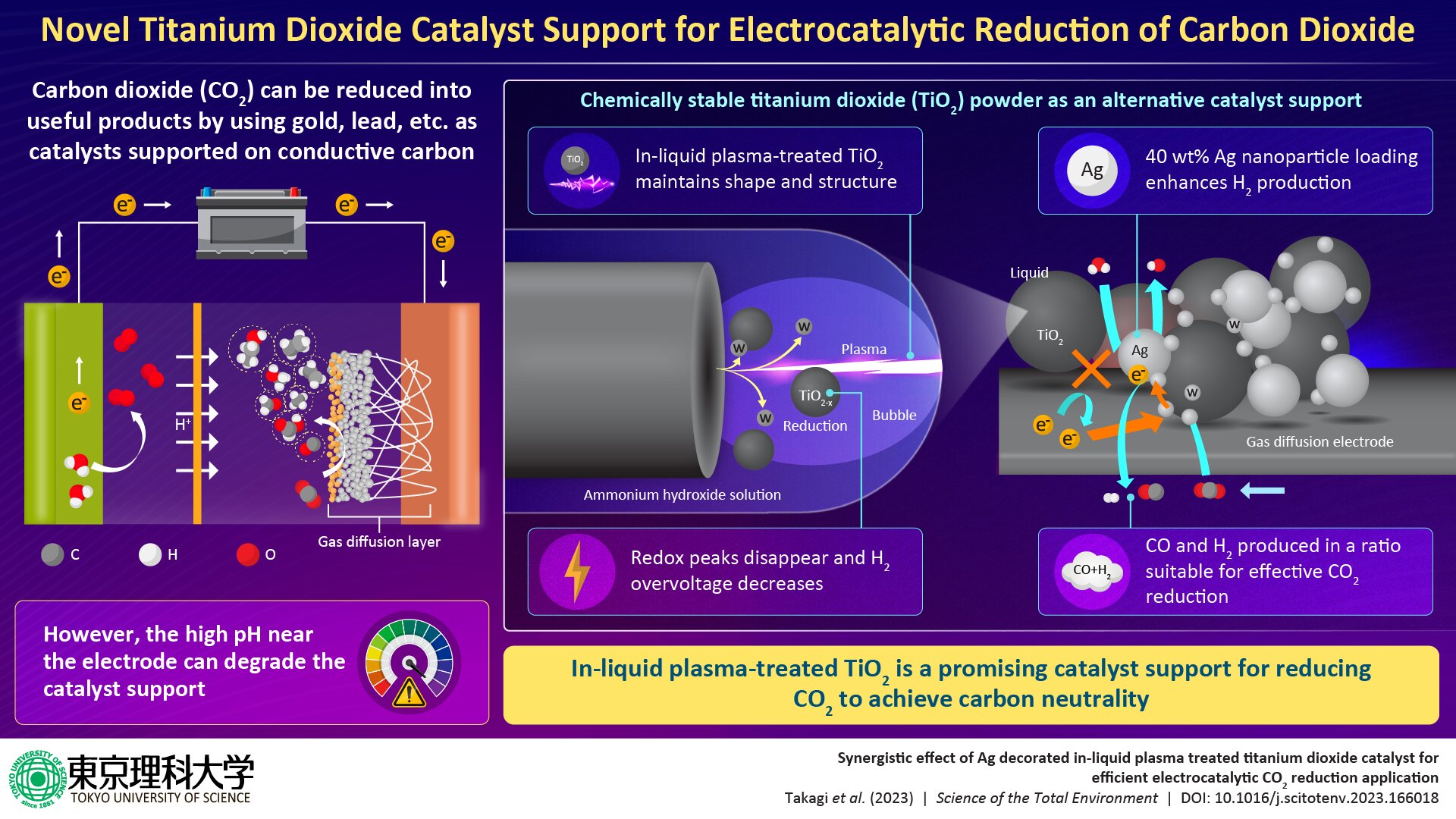
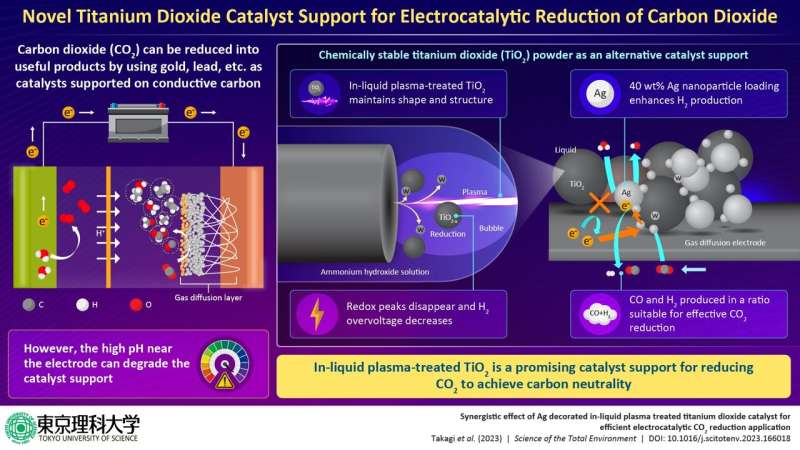
The conversion of atmospheric carbon dioxide (CO2), a greenhouse gas, to useful resources such as carbon monoxide, formic acid, and methanol and their byproducts is considered a promising route to mitigating global warming as well as generating economic value. One approach to CO2 conversion is through electrocatalytic reduction.
This process utilizes conventional catalysts, such as lead, silver, tin, copper, gold etc. supported on conductive carbon as electrode material for selectively CO2 reduction. However, the electrode is often exposed to a high pH environment of the electrolyte during electrocatalysis, which can degrade the catalyst support and is a cause of major concern.
To address this challenge, a team of researchers, led by Mr. Kai Takagi and Prof. Chiaki Terashima from Graduate School of Science and Technology and Research Institute for Science and Technology at Tokyo University of Science (TUS) in Japan, has recently developed a catalyst support based on titanium dioxide (TiO2) powder, a compound commonly used in sunscreen, paints, coatings, toothpaste, plastics, paper, pharmaceuticals, and food coloring, as an alternative to carbon for facilitating effective CO2 reduction.
Their work was published in Science of the Total Environment.
The researchers first carried out surface treatment using safe and inexpensive in-liquid plasma to improve the electrochemical properties of TiO2.
“The in-liquid plasma-treated TiO2 maintained its particle shape and crystal structure. Additionally, elemental analysis and evaluation of the interfacial bonding state and electrochemical properties of TiO2 revealed that the redox peaks corresponding to Ti4+ and Ti3+ derived from TiO2 disappeared and the hydrogen overvoltage decreased,” said Prof. Terashima.
These observations led the team to conclude that tungsten coating or doping occurred on some portions of the reduced TiO2 surface.
The researchers then used the TiO2 as a carrier and loaded it with silver nanoparticles (AgNPs), which act as catalysts, to develop a gas diffusion electrode for CO2 reduction. While untreated TiO2 exhibited high selectivity for CO2 and carbon black, in-liquid plasma-treated TiO2 with 40 wt% AgNP loading demonstrated increased hydrogen production and enhanced catalytic performance.
Given that a suitable ratio of hydrogen to carbon monoxide is important for effective CO2 reduction, the presented technology, thus, has tremendous potential for converting CO2 to useful byproducts, such as syngas, which is considered a clean fuel with very high industrial value.
Additionally, the electrocatalytic reduction of CO2 can be integrated with renewable energy sources, such as solar panels or wind power, for sustainable and environmentally friendly CO2 conversion. Therefore, this work is a significant step towards efficiently tackling greenhouse gas emissions and fighting climate change.
“Hopefully, the present study will promote research on technologies for carbon neutrality and carbon recycling, in alignment with the United Nations Sustainable Development Goals 7, 12, and 13 on affordable and clean energy, responsible consumption and production, and climate action, respectively. These, in turn, will open doors to the realization of a carbon-neutral and sustainable future,” concludes Prof. Terashima.
More information:
Kai Takagi et al, Synergistic effect of Ag decorated in-liquid plasma treated titanium dioxide catalyst for efficient electrocatalytic CO2 reduction application, Science of The Total Environment (2023). DOI: 10.1016/j.scitotenv.2023.166018
Provided by
Tokyo University of Science
Citation:
Novel titanium dioxide catalyst shows promise for electrocatalytic carbon dioxide reduction (2023, September 5)
retrieved 5 September 2023
from https://phys.org/news/2023-09-titanium-dioxide-catalyst-electrocatalytic-carbon.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.