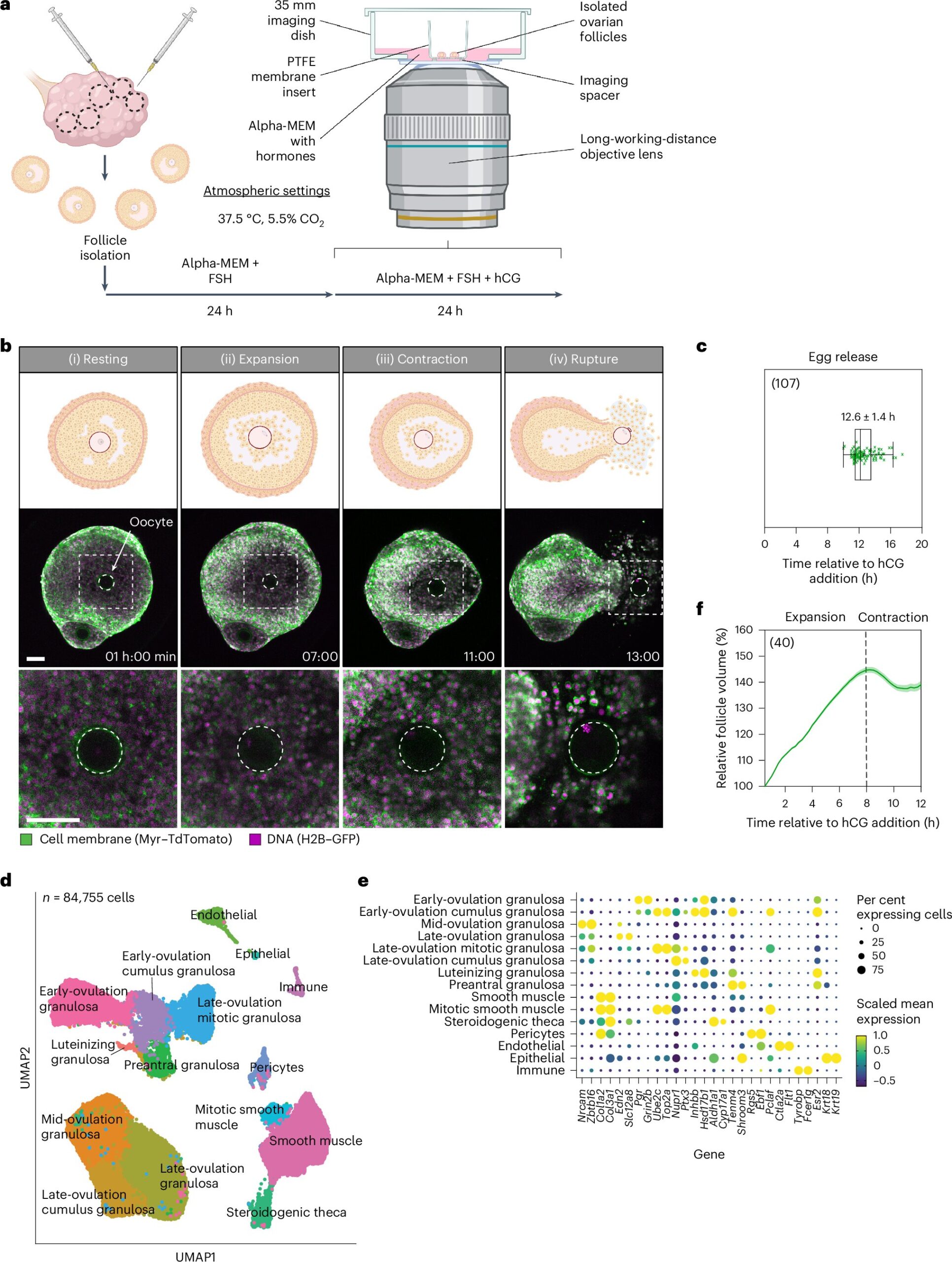
Researchers at the Max Planck Institute for Multidisciplinary Sciences have developed a live imaging method that reveals the process of ovulation in unprecedented detail. The team followed initial observations by performing inhibition testing to confirm causal effects.
Oocytes are immature eggs stored in ovarian follicles. Once per ovarian cycle, an oocyte matures into a fertilizable egg and is released from the follicle. This release process is what we call ovulation.
Ovulation studies are usually done with fixed tissues, capturing set time points of the process. While useful, these point-in-time analyses are limited when trying to understand something as dynamic as ovulation.
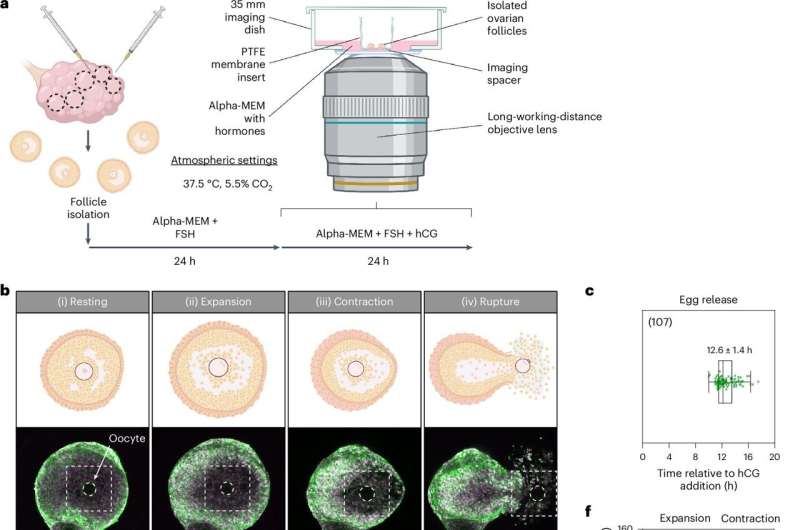
The technical report, “Ex vivo imaging reveals the spatiotemporal control of ovulation,” published in Nature Cell Biology, shows how researchers used a new image capture method to identify three distinct phases of ovulation: follicle expansion, contraction, and rupture, culminating in the release of an egg.
Images were captured using a combination of confocal and two-photon microscopy, live imaging isolated mouse ovarian follicles. Transgenic mouse donors with expression markers for cell membranes and DNA helped make intricate movements visible.
The technique captured the ovulation process by imaging at 10-minute intervals over 24 hours, allowing detailed observation of ovulation at both the cellular and whole follicle levels. The team followed initial observations and data collection results by performing inhibition testing to confirm causal effects.
The study identified follicle expansion as the first phase, driven by hyaluronic acid secretion and fluid influx, which increased follicle volume. Initial observations of increases led to an inhibition test of hyaluronic acid synthesis, resulting in reduced expansion and blocked ovulation, confirming the direct role.
Next came contraction, controlled by smooth muscle cells in the outer follicle layer, possibly regulated by progesterone and endothelin signaling. Inhibition of these pathways reduced contraction and ovulation rates, confirming the interplay.
Finally, follicle rupture began, followed by the release of follicular fluid, cumulus cells, and the egg. Rupture and egg release were seen to occur in three steps: fluid rupture, cellular rupture, and egg release.
Molecular mechanisms of mitochondrial energy production and actomyosin contractility were also found to drive the process. Between 6 and 9 hours after triggering initial ovulation, gene expression analysis revealed an upregulation of pathways related to mitochondrial respiratory chain activity, indicating a high demand for ATP in smooth muscle cells during contraction. Inhibition of mitochondrial function impaired contraction and prevented ovulation, demonstrating the reliance on mitochondrial ATP production.
Additionally, the researchers identified an increase in the expression of genes related to actomyosin contractility between 9 and 12 hours post-hCG. Blocking actomyosin contractility further confirmed the necessity of actomyosin-driven contraction for follicle rupture and egg release.
This new approach not only uncovers detailed spatiotemporal patterns of ovulation and opens new avenues for investigating how the cellular and molecular mechanisms interdepend in reproduction, but provides truly amazing video of nature at work.
More information:
Christopher Thomas et al, Ex vivo imaging reveals the spatiotemporal control of ovulation, Nature Cell Biology (2024). DOI: 10.1038/s41556-024-01524-6
© 2024 Science X Network
Citation:
Live imaging of ovulation in action reveals three distinct phases (2024, October 22)
retrieved 22 October 2024
from https://phys.org/news/2024-10-imaging-ovulation-action-reveals-distinct.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.