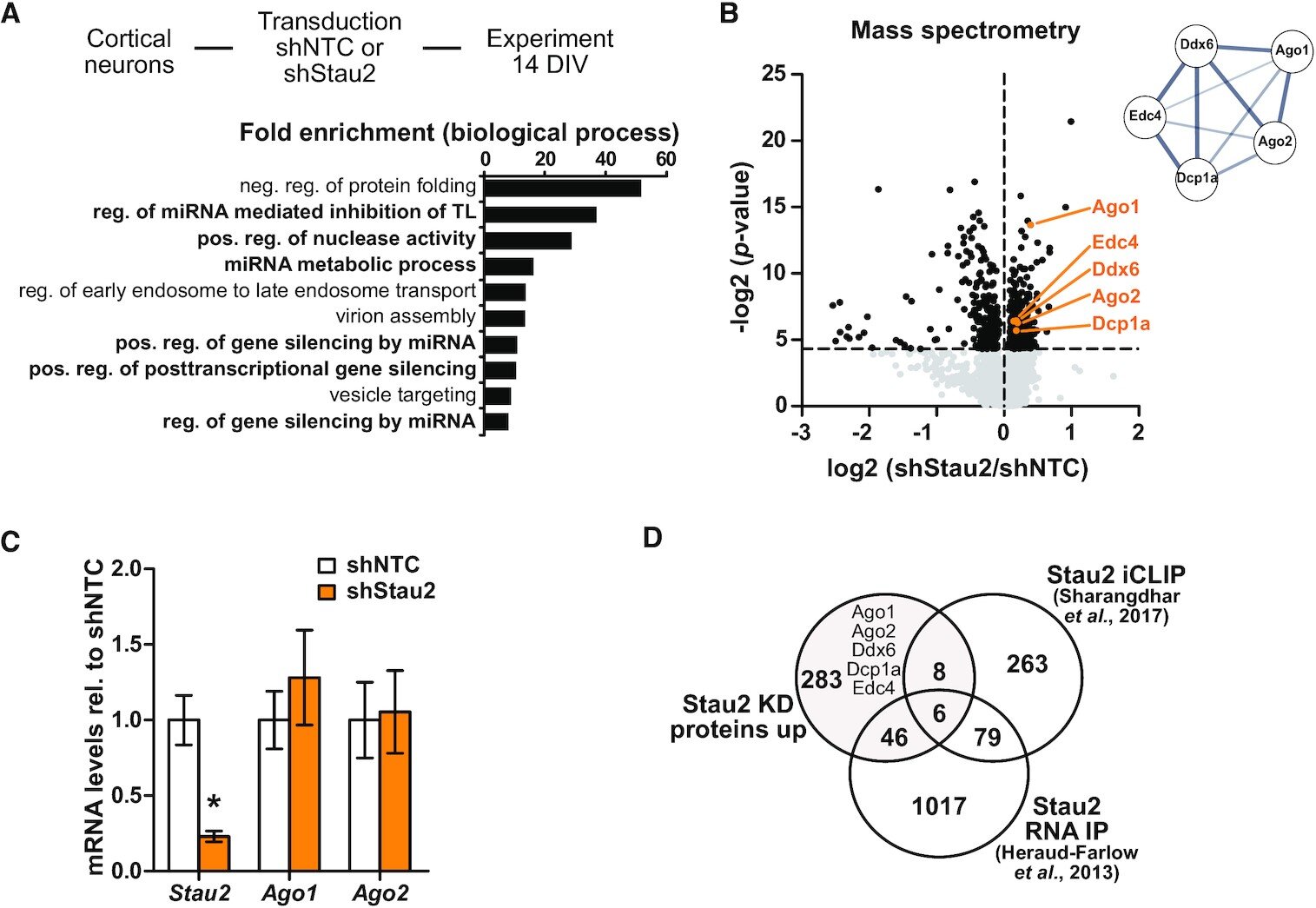
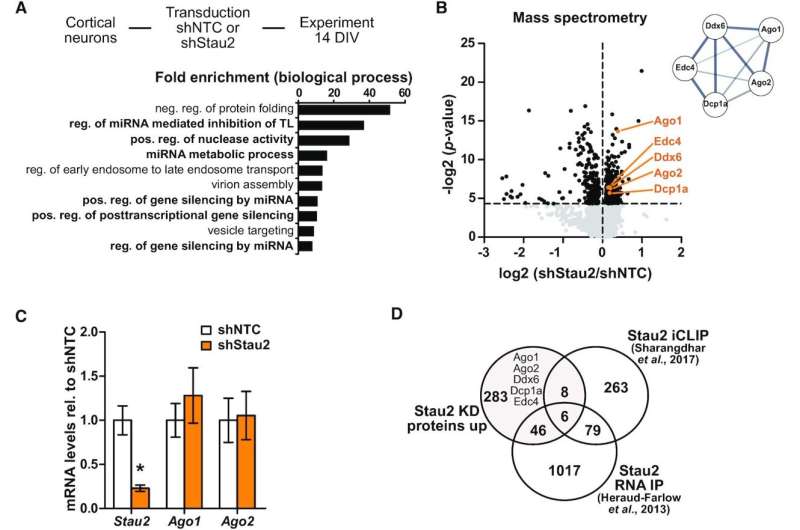
Neurons constantly adapt to new requirements. This plasticity is the molecular foundation of learning and remembering. At the cellular level, there is a variety of mechanisms for regulating general gene expression.
One of the major players is RNA-binding proteins, which recognize messenger molecules (mRNA). In this way, they regulate where and when proteins can be produced inside the neuron. Jointly with other components, the Staufen2 and Argonaute RNA-binding proteins form RNA granules in the cytoplasm.
A team led by LMU cell biologist Prof. Michael Kiebler has now shown for the first time how Staufen and Argonaute proteins interact with each other.
The authors of the study published in Nucleic Acids Research were able to demonstrate that the two RNA-binding proteins compete with each other in fulfilling their function.
Their results suggest that in this way the two RNA-binding proteins regulate the translation of specific proteins in the dendrite and at the synapse.
The scientists hypothesize that these assembly dynamics of RNA granules make an important functional contribution to synaptic plasticity, particularly in neurons.
Small aggregates function as temporary RNA repositories, regulated by neural activity
Janina Ehses et al, The dsRBP Staufen2 governs RNP assembly of neuronal Argonaute proteins, Nucleic Acids Research (2022). DOI: 10.1093/nar/gkac487
Ludwig Maximilian University of Munich
Citation:
Exploring how Staufen and Argonaute proteins interact with each other (2022, July 5)
retrieved 6 July 2022
from https://phys.org/news/2022-07-exploring-staufen-argonaute-proteins-interact.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.